  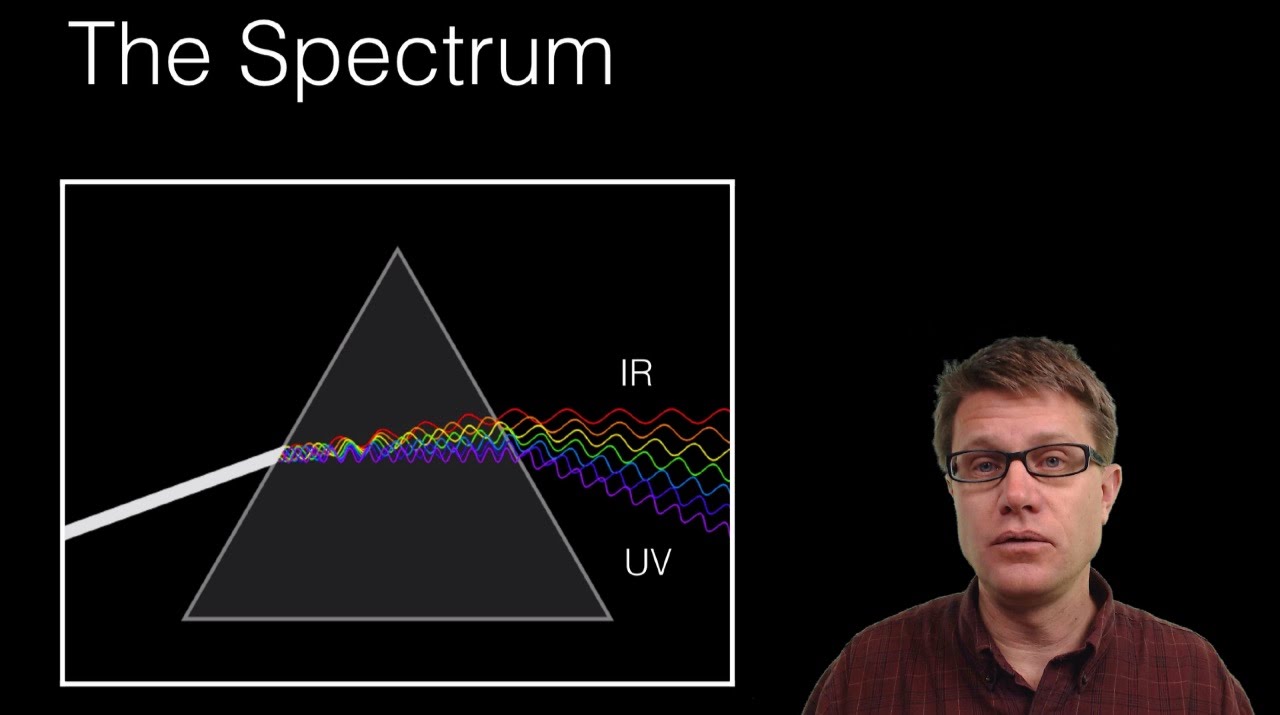
Each line reflects the unique properties of the elements. These spectra are emitted by agitated molecules or ions. This spectrum has sharp lines with finite wavelengths or frequencies. The line spectrum can also be called the not continuous spectrum. The line spectrum is obtained when high-temperature gas is passed through a prism. The emission spectrum can be divided into three types. Each light source has a unique emission spectrum. The spectrum derived from the luminous source is the self-luminous emission spectrum. The spectrum can be divided into two types, the emission spectrum, and the absorption spectrum. When it comes to more than one spectrum it is called the electromagnetic spectrum. The spectrum is the set of colors obtained on the screen by the color spectrum. The name for this process is the color spectrum of light. When white light passes through a prism it can be calculated as a continuous spectrum on a screen that splits into seven colors. Also glows white when heated.įig:1 Emission spectra What are Emission Spectra? And the object glows a pure orange when heated continuously. When an object is heated, it begins to glow red as it warms up. That is, the object emits electromagnetic radiation. The energy of the electromagnetic wave is from the energy of the oscillating electrons available. The speed of light in a medium is less than the speed of light in a vacuum. The ratio of the electric and magnetic fields in any medium is equal to the velocity of the electromagnetic wave propagating in that medium. When an electron oscillates with a focal point (or molecular dipole oscillates), it generates electromagnetic waves. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
